Expert Laboratory Billing Services | 30+ Years of Lab RCM
Laboratory Billing Services That Help You Tackle Revenue Cycle Management Challenges.
Schedule Your Consultation Today
Maximize the reimbursement process and streamline laboratory operations
PGM understands that clinical and reference laboratories face a daunting number of challenges. The complexity of clinical laboratory billing, ever-increasing regulatory demands, inadequate legacy applications and difficulties accessing information can impede growth and hinder the success of your business.
PGM has been working with independent clinical, hospital outreach and reference labs for the past 30 years and has developed one of the most effective lab billing, coding and financial management services on the market today. With PGM, clinical and reference labs can effectively manage high-volume, small-dollar claims, improve laboratory collection rates and gain revenue cycle management insight through in-depth financial management reports.
Every step of the way, our team works closely with you to ensure we develop a customized billing strategy that is designed to meet the strategic business objectives of your laboratory. Our full-service laboratory billing handles all aspects of laboratory billing, including both the technical and professional components, both in-network and out of network billing. In addition PGM services include:
- Collection of payer payments made to directly to patients
- LIS Integration and data integration
- Sales Representative Tracking at the collection level
- Accession or patient encounter monitoring to ensure every case is submitted for reimbursement
- Monitor payments to ensure adherence to contracted rates
- Appeals for molecular laboratory claims denied as investigational
- Patient balance billing
- Handle patient inquiries regarding billing questions
- Guidance on pricing
- Ensure accurate financial reporting, coding, billing and collections
- Increase revenue while cost-effectively processing high-volume, low-dollar claims
- Regulatory guidance
To learn more about our laboratory medical billing services, contact us for a free consultation.
Laboratory Revenue Cycle Management
Improved Performance
Focus on growing your laboratory, not managing your billing. Our competitive laboratory billing service is focused on maximizing your collections, getting you paid more and paid faster. Our advanced claim submission reviewer detects lab billing errors prior to entry, dramatically reducing denial rates. We aggressively follow-up and appeal denials and misadjudicated claims, increasing your overall revenue. Specifically, you will improve your collection rates and reimbursement cycle, reduce your accounts receivable, decrease the rate of lost and denied claims and gain improved insight into your laboratory's financials and performance. With PGM, you will have 100% visibility over your claims, and you will know where your cash is.
By letting PGM manage the task of collecting payments and monitoring reimbursement, you and your staff will have more time to focus on your success and growth.
Personalized Assistance
Our staff is 100% committed to serving you. All of our clients are assigned their own designated account executive, giving you the security and comfort of working with a representative who is uniquely familiar with your business. From guiding you through the implementation process to frequent performance reviews and updates, our staff is there with you every step of the way. In addition PGM's Lab Intelligence Team monitors the frequent changes in the industry, providing you with invaluable industry intelligence, helping to guide you on policy issues and regulatory compliance.
Transparency
Understanding your laboratory's financial performance can be challenging. With data buried in spreadsheets and antiquated systems, gathering the information necessary to optimize operations and plan for the future is often difficult and time consuming.
To help meet this challenge, PGM provides you with advanced financial and practice analysis tools, specifically designed to give enhanced visibility of your operations and capture key data and metrics for internal benchmarking and regulatory reporting. Measure everything from sales representative performance to accession summary data. PGM's cloud based laboratory billing system is a robust tool trusted by laboratories across the country.
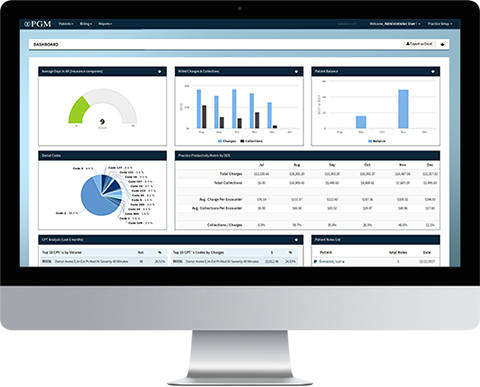
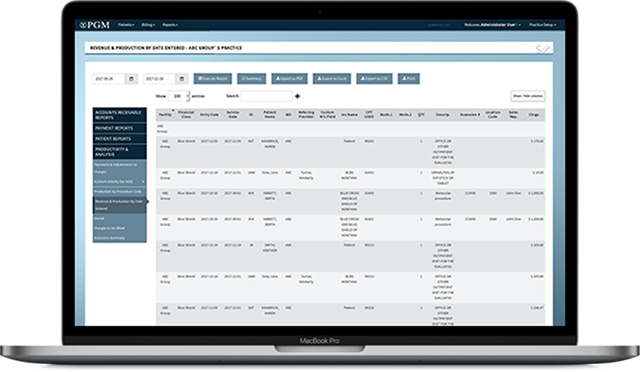
Reporting
PGM laboratory billing reporting is highly intuitive and grants instant access to data so you can immediately track performance and make qualified decisions. Qualitative assessment becomes easier because you can review everything from which payers reimburse, and at what rates to which business lines are most successful. Additionally, you will have full insight into specimen origin data and track referral sources at both the accession summary and detail level. Our robust reporting engine provides data on nearly any field or metrics, and is available in real time from any internet enabled device.
See our solutions in action
PGM's Free Medical Billing Software Demo
Please complete the following form and a PGM representative will contact you.
Contact us directly at 877-224-6206 to schedule a live demo.